Otoscopio de Fibra Óptica
How to Import Medical Equipment from China: Step-by-Step Guide for Hospitals and Distributors
The Opportunity — and the Complexity
China is the world's largest manufacturer of medical equipment. From diagnostic imaging systems and patient monitors to dialysis machines and surgical lighting, Chinese manufacturers produce hospital-grade equipment at price points that can run 30–60% lower than comparable Western brands — without necessarily sacrificing clinical performance.
For hospitals expanding their departments, clinics equipping new facilities, and distributors building out their product lines, sourcing from China makes strong financial sense. But the import process is not simple. Regulatory compliance, supplier verification, customs documentation, and logistics all have to work together before a single piece of equipment reaches your facility.
This guide walks through every stage of that process — practically and clearly, without glossing over the parts that tend to go wrong.
Step 1: Define Exactly What You Need Before You Source Anything
This sounds obvious. Many procurement teams skip it anyway, jumping straight to supplier browsing — and that creates problems downstream.
Before contacting any supplier or requesting a quote, document:
- The clinical purpose of the equipment — what procedures it will support, what patient volume it needs to handle
- Technical specifications — power requirements (voltage/frequency), dimensions, connectivity standards, software language
- Regulatory certifications required in your country — CE marking for Europe, FDA clearance for the US, ANVISA for Brazil, and so on
- Quantity and delivery timeline
- Budget range, including realistic estimates for shipping, import duties, and local installation
Getting this right upfront means you can evaluate suppliers accurately, compare quotes on equal terms, and avoid ordering equipment that fails your local compliance requirements.
Step 2: Understand the Regulatory Landscape
This is where most first-time importers underestimate the complexity. Medical devices are regulated products in every major market. Importing equipment without the required certifications can result in customs seizure, significant fines, or equipment that cannot legally be used in a clinical setting.
CE Marking (Europe and Many Developing Markets)
CE marking indicates that a device meets EU safety and performance standards. Many countries outside Europe — including those in the Middle East, Africa, and Southeast Asia — accept CE-marked devices as a baseline compliance standard. When sourcing from Chinese manufacturers, always verify that the CE certificate is genuine and current. Request the full certificate, not just a logo on a brochure.
FDA Registration (United States)
Importing medical devices into the US requires FDA registration. Most devices fall under Class I, II, or III classification. Class II devices — which cover a large proportion of diagnostic and monitoring equipment — typically require 510(k) clearance. Some Chinese manufacturers have obtained FDA registration for their products (Mindray, for example, has a substantial FDA-cleared portfolio), but many have not. If you are importing into the US, this is non-negotiable.
ISO 13485
This is the international quality management standard for medical device manufacturers. It is not a market access certification in itself, but ISO 13485 certification signals that a manufacturer's production and quality control processes meet a recognized international standard. Treat it as a baseline requirement when vetting suppliers.
Local Market Registrations
Beyond CE and FDA, many countries require their own device registration before import is permitted — India (CDSCO), Brazil (ANVISA), Saudi Arabia (SFDA), and others. These processes can take months. If you are importing into a market with its own device registry, factor that timeline into your procurement plan early, or work with a supplier or distributor who already holds local registration for the products you need.
Step 3: Vet Your Suppliers Rigorously
The Chinese medical equipment market spans a wide spectrum — from globally recognized manufacturers with decades of clinical track record to small factories producing equipment of uncertain quality. Supplier selection needs to be thorough.
What to Look For
Manufacturing credentials: Are they the actual manufacturer or a trading company? Both can be legitimate, but you need to know which you are dealing with. Manufacturers generally offer more transparency into production quality and tend to provide stronger after-sales support.
Certifications on file: Request copies of CE certificates, ISO 13485 certification, and any FDA registration documents. Verify these independently where possible — CE certificates can be checked against notified body databases.
Export experience: Has the supplier exported to your specific country before? Do they understand your local import requirements? A supplier with established export channels to your region will handle documentation and compliance more reliably.
References: Ask for references from other hospital or distributor clients. Reputable suppliers will have them. If a supplier cannot provide any verifiable references, that is worth taking seriously.
After-sales support: Medical equipment requires installation, commissioning, calibration, and ongoing maintenance. Clarify what the supplier provides post-delivery — warranty terms, spare parts availability, remote technical support, and whether they have service partners in your country.
Established Chinese Manufacturers Worth Knowing
Several Chinese manufacturers have built strong international reputations and are useful benchmarks when evaluating the market:
- Mindray — patient monitoring, ultrasound, in-vitro diagnostics; widely FDA-cleared and CE-marked
- Sonoscape — ultrasound systems with a strong presence in emerging markets
- Edan Instruments — ECG, patient monitoring, ultrasound
- Neusoft Medical — CT and MRI systems
- United Imaging — advanced imaging including PET-CT
These manufacturers distribute through authorized channels globally. Platforms like China Care Medical aggregate products from these and other established manufacturers, making it easier to browse, compare, and initiate inquiries without navigating each manufacturer's sales process individually.
Step 4: Request and Evaluate Quotations
With a shortlist in hand, request formal quotations. A proper quote for medical equipment should include:
- Unit price and total price
- Incoterms (more on this below)
- Payment terms
- Lead time from order confirmation to shipment
- Warranty period and terms
- Included accessories and consumables
- Packaging and shipping specifications
- Applicable certifications
Understanding Incoterms
Incoterms define who is responsible for freight, insurance, and risk at each stage of the shipment. For medical equipment imports, the most common terms are:
Incoterm | What It Means for the Buyer |
EXW (Ex Works) | You arrange and pay for everything from the factory gate. Maximum control, maximum responsibility. |
FOB (Free on Board) | Supplier delivers to the port of origin and loads onto the vessel. You handle ocean freight, insurance, and import. |
CIF (Cost, Insurance, Freight) | Supplier arranges and pays for freight and insurance to your destination port. You handle import clearance and inland delivery. |
DDP (Delivered Duty Paid) | Supplier handles everything, including import duties. Simplest for the buyer, but with the least visibility and control. |
For most hospital and distributor buyers, FOB or CIF offers a reasonable balance — you maintain visibility over the shipment while the supplier handles Chinese export logistics. DDP can work for smaller orders where simplicity outweighs cost.
Step 5: Negotiate and Formalize the Contract
Do not proceed on a purchase order alone. For medical equipment, a formal sales contract is essential. It should cover:
- Detailed product specifications (model numbers, configurations, software versions)
- Price, currency, and payment schedule
- Delivery timeline and penalties for delay
- Inspection rights before shipment
- Warranty terms and scope
- Dispute resolution mechanism and governing jurisdiction
- Intellectual property and confidentiality clauses where relevant
Payment terms for first-time transactions typically follow a 30% deposit on order confirmation, with the 70% balance due before shipment or against shipping documents. As you build a relationship with a supplier, terms may become more flexible. Avoid paying 100% upfront with an unverified supplier.
Pre-Shipment Inspection
For significant orders, arrange a third-party pre-shipment inspection. Companies like SGS, Bureau Veritas, and Intertek provide this service. An inspector visits the factory before goods are loaded, verifies that the equipment matches your specifications, checks packaging, and confirms quantities. The cost is modest relative to the risk of receiving incorrect or damaged equipment.
Step 6: Navigate Export Documentation from China
Your supplier handles Chinese export procedures, but you need to ensure they provide the right documentation. Standard documents for medical equipment exports from China include:
- Commercial Invoice — itemized, with accurate declared value
- Packing List — detailed breakdown of contents, weights, and dimensions
- Bill of Lading or Airway Bill — issued by the freight carrier
- Certificate of Origin — often required for preferential tariff treatment under trade agreements
- CE Certificate / FDA Registration Certificate — copies for customs and regulatory purposes
- Export License (if applicable) — some medical devices require Chinese export licensing
Confirm with your supplier which documents are required for your specific destination. Missing or incorrect documentation is one of the most common causes of customs delays.
Step 7: Choose Your Shipping Method
Medical equipment moves by sea freight, air freight, or — for smaller items — express courier. Each has trade-offs.
Sea Freight
The standard choice for large or heavy equipment: imaging systems, operating tables, dialysis machines. Transit times from Chinese ports to major global destinations typically range from 15 to 45 days depending on routing, and sea freight is significantly cheaper per kilogram than air.
For fragile or high-value equipment, specify FCL (Full Container Load) where possible. This reduces the handling and co-loading risks that come with LCL (Less than Container Load) shipments.
Air Freight
Used for urgent orders, smaller devices, or high-value items where the cost of delay outweighs the freight premium. Patient monitors, portable ultrasound units, and diagnostic devices are commonly shipped by air. Expect transit times of 3–7 days.
Freight Forwarder
Whether you ship by sea or air, use a freight forwarder with medical equipment experience. They handle booking, documentation, customs coordination, and delivery. A good forwarder who knows your destination market's import requirements is worth their fee.
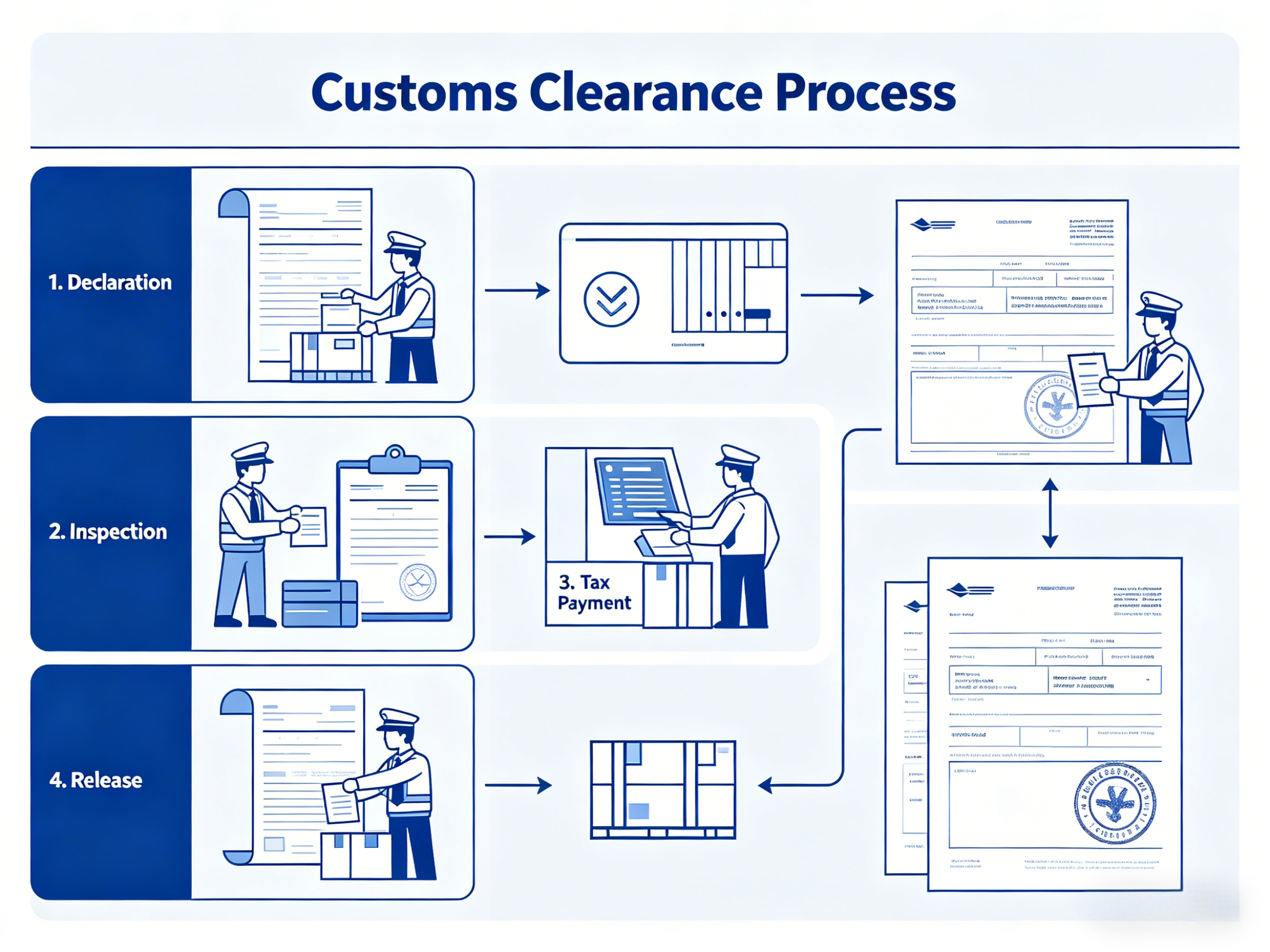
Step 8: Clear Customs at Your End
Import customs clearance for medical devices requires attention to both classification and compliance.
HS Codes
Every imported product is classified under a Harmonized System (HS) code. Medical equipment has specific HS codes, and getting this right matters — it determines the applicable import duty rate. Misclassification can mean overpaying duty or triggering a customs query.
Import Duties and Taxes
Duty rates on medical equipment vary by country and product type. Many countries apply reduced or zero duty rates on medical devices to encourage healthcare investment, but this varies considerably. Your freight forwarder or customs broker can advise on applicable rates and any available exemptions.
Regulatory Clearance at Import
Some countries require import permits or regulatory approval before medical devices can clear customs — separate from any CE or FDA certification the device already carries. In markets with their own device registration systems (India, Brazil, Saudi Arabia, and others), you may need to present local registration documentation at the border. If that is not secured in advance, the shipment can be held.

Step 9: Installation, Commissioning, and Training
Medical equipment does not simply arrive and start working. Most clinical devices require:
- Professional installation — particularly for imaging equipment, operating room systems, and dialysis units, which may need infrastructure preparation covering electrical, plumbing, or ventilation
- Commissioning and calibration — confirming that the equipment performs to specification
- User training — clinical and technical staff need to be trained on operation and basic maintenance
- Local regulatory compliance — in some markets, equipment must be registered with local health authorities before clinical use
Clarify with your supplier what installation and training support they provide. Some manufacturers send engineers for on-site commissioning; others offer remote support or detailed technical documentation that allows local biomedical engineers to handle installation.
If you are procuring a complete department setup — a dialysis center, an operating room, a radiology suite — a turnkey project approach can simplify this considerably. Suppliers experienced in turnkey hospital projects coordinate equipment supply, installation, and commissioning as a single package.
Step 10: Establish Ongoing Support and Spare Parts Access
The total cost of owning medical equipment includes maintenance, calibration, and eventual parts replacement. Before finalizing any purchase, understand:
- Warranty period — typically 12–24 months for most equipment
- What the warranty covers — parts, labor, on-site service, or remote support only
- Spare parts availability — can you source parts locally, or do they need to be imported from China? What are typical lead times?
- Service contracts — does the manufacturer or a local partner offer post-warranty service agreements?
This matters most for complex equipment like CT scanners, MRI machines, or dialysis systems, where downtime has direct clinical consequences.
Common Mistakes to Avoid
Choosing on price alone. The cheapest quote often reflects corners cut on quality control, certification, or after-sales support. Evaluate total cost of ownership, not just the purchase price.
Skipping the compliance check. Never assume a device is compliant because the supplier says so. Request certificates and verify them independently.
Underestimating lead times. Manufacturing, shipping, customs clearance, and installation can collectively take 3–6 months for complex equipment. Plan accordingly.
Ignoring local registration requirements. If your country requires device registration before import, this needs to be resolved before you place the order — not after the shipment arrives.
No pre-shipment inspection. For large orders, this is straightforward insurance against receiving something that does not match what you ordered.
Where China Care Medical Fits Into This Process
Navigating the Chinese medical equipment market is considerably easier when the supplier aggregation work has already been done for you. China Care Medical brings together equipment from established Chinese manufacturers — organized by clinical department, from imaging and operating rooms to dialysis and ICU — so hospitals, clinics, and procurement teams can browse, compare, and submit inquiries without approaching each manufacturer separately.
For organizations looking at turnkey department setups — laminar flow operating rooms, haemodialysis centers, and similar projects — the platform also showcases completed projects that illustrate what full-scale procurement and installation looks like in practice.
Final Thoughts
Importing medical equipment from China is a legitimate, well-established procurement strategy for hospitals and distributors worldwide. The manufacturers are real, the quality can be excellent, and the cost advantages are significant. But the process requires discipline — on specifications, compliance, supplier vetting, documentation, and logistics.
The teams that do this well treat it as a professional procurement process, not a shortcut. They verify certifications, use proper contracts, inspect before shipment, and plan for installation and ongoing support from day one.
If you are in the early stages of planning a procurement or exploring Chinese suppliers for the first time, start by getting clear on your specifications and your local regulatory requirements. Everything else follows from there.
Learn more at chinacaremedical.com